Safe Start
Improving Child Survival through the Evaluation of EPI Interventions in Sierra Leone
- Duración
- 15/03/2025 - 15/09/2026
- Coordinador
- Clara Menéndez
- Financiadores
- Fundación ”la Caixa”
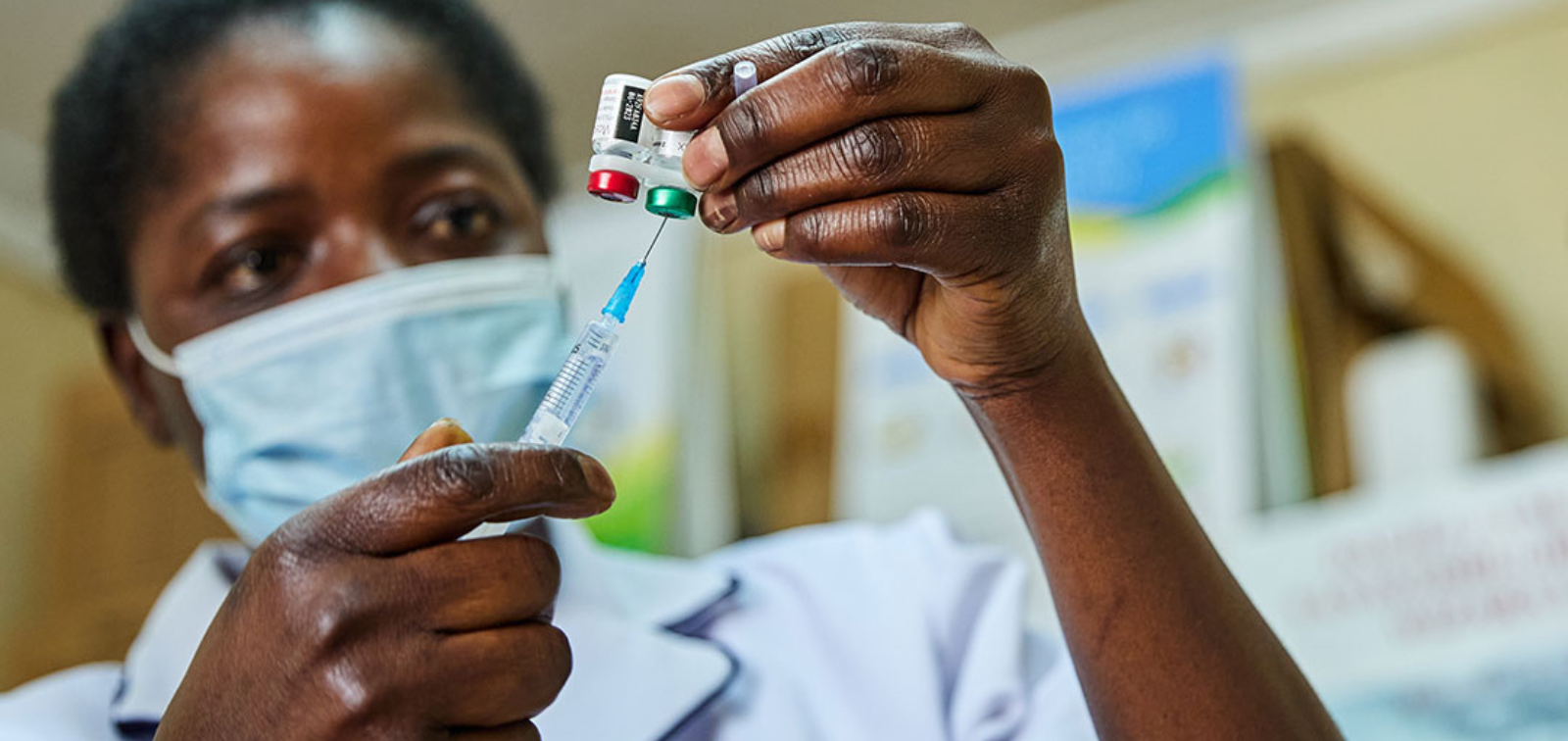
Malaria remains a leading cause of illness and death among young children in Sierra Leone, despite existing prevention and treatment efforts. In 2024, the country became one of the first in Africa to introduce the RTS,S malaria vaccine into its Expanded Program on Immunization (EPI), marking a significant step in the fight against the disease. However, questions remain about how best to integrate the vaccine into routine health services and ensure its accessibility for all children.
SafeStart is a pioneering initiative designed to evaluate the real-world implementation and impact of the RTS,S malaria vaccine in Sierra Leone. Through a combination of community surveys, health system data analysis, and stakeholder engagement, the project aims to:
- Assess vaccine coverage, uptake, and effectiveness in reducing malaria burden. Identify barriers and facilitators to vaccine acceptance and delivery at the community and health system levels.
- Explore how integrating RTS,S with other childhood immunizations and preventive treatments can enhance health outcomes.
- Provide evidence-based recommendations to strengthen immunization programs and child health policies.
- Train healthcare workers to enhance their capacity in vaccine delivery, monitoring, and integration, using a co-designed curriculum with the Ministry of Health.
By generating critical insights, SafeStart will support the Ministry of Health and global health partners in ensuring that the malaria vaccine reaches the children who need it most. Ultimately, this project is about improving child survival, enhancing healthcare delivery, and shaping the future of malaria prevention in Sierra Leone and beyond.
Total Funding
400.000€
Nuestro equipo
Coordinación
-
 Clara Menéndez Research Professor, Directora de la Iniciativa y del Programa de Salud Materna, Infantil y Reproductiva
Clara Menéndez Research Professor, Directora de la Iniciativa y del Programa de Salud Materna, Infantil y Reproductiva
Equipo ISGlobal
-
Maya Sibley Project manager
-
 Cristina Enguita Assistant Research Professor
Cristina Enguita Assistant Research Professor -
 Cristina Raya Gambus Project Assistant
Cristina Raya Gambus Project Assistant -
-
 Andreu Bofill Data Manager
Andreu Bofill Data Manager
Otros proyectos
Ver proyectos pasadosCaDMIA-plus
Validación continuada de la autopsia mínimamente invasiva (MIA) para la investigación de la causa de muerte en niños pequeños, y desarrollo de un centro de investigación y formación sobre el estudio de la causa de muerte
MAMAH
Improving Maternal and Infant Health by reducing malaria risks in African women: evaluation of the safety and efficacy of dihydroartemisinin-piperaquine for intermittent preventive treatment of malaria in HIV-infected pregnant women
MIBio
Identification of Prematurity and Pre-Eclampsia as Causes of Mortality
MA-CoV
Prevalence and impact of SARS-CoV-2 infection on maternal and infant health in African populations
IPERVAC-SL
Impact of perceptions of COVID-19 vaccines on health-seeking behaviours in Sierra Leone
RESPIRE
Respiratory syncytial virus (RSV) in African pregnant women and children
VITAL
Towards preparedness for new maternal vaccinations: Understanding barriers and facilitators to maternal vaccine acceptance
SARA (SARS-CoV-2 and Acetylsalicylic acid)
Efficacy of low dose acetylsalicylic acid in preventing adverse maternal and perinatal outcomes in SARS-CoV-2 infected pregnant women
HIV drug resistance (HIVDR)
Description of HIV drug resistance patterns and its association with the risk of HIV mother to child transmission among pregnant women from southern Mozambique
Evaluación de la eficacia del ácido acetilsalicílico a dosis bajas en la prevención de los efectos maternos y perinatales adversos en mujeres embarazadas infectadas por el SARS-CoV-2
PROTECT
PReparing for Optimal Phase III/IV maTErnal Group B StreptococCal vaccine Trials in Africa
ICARIA
Improving Care through Azithromycin Research for Infants in Africa
MULTIPLY
MULTIple doses of IPTi Proposal: a Lifesaving high Yield intervention




